Fentanyl
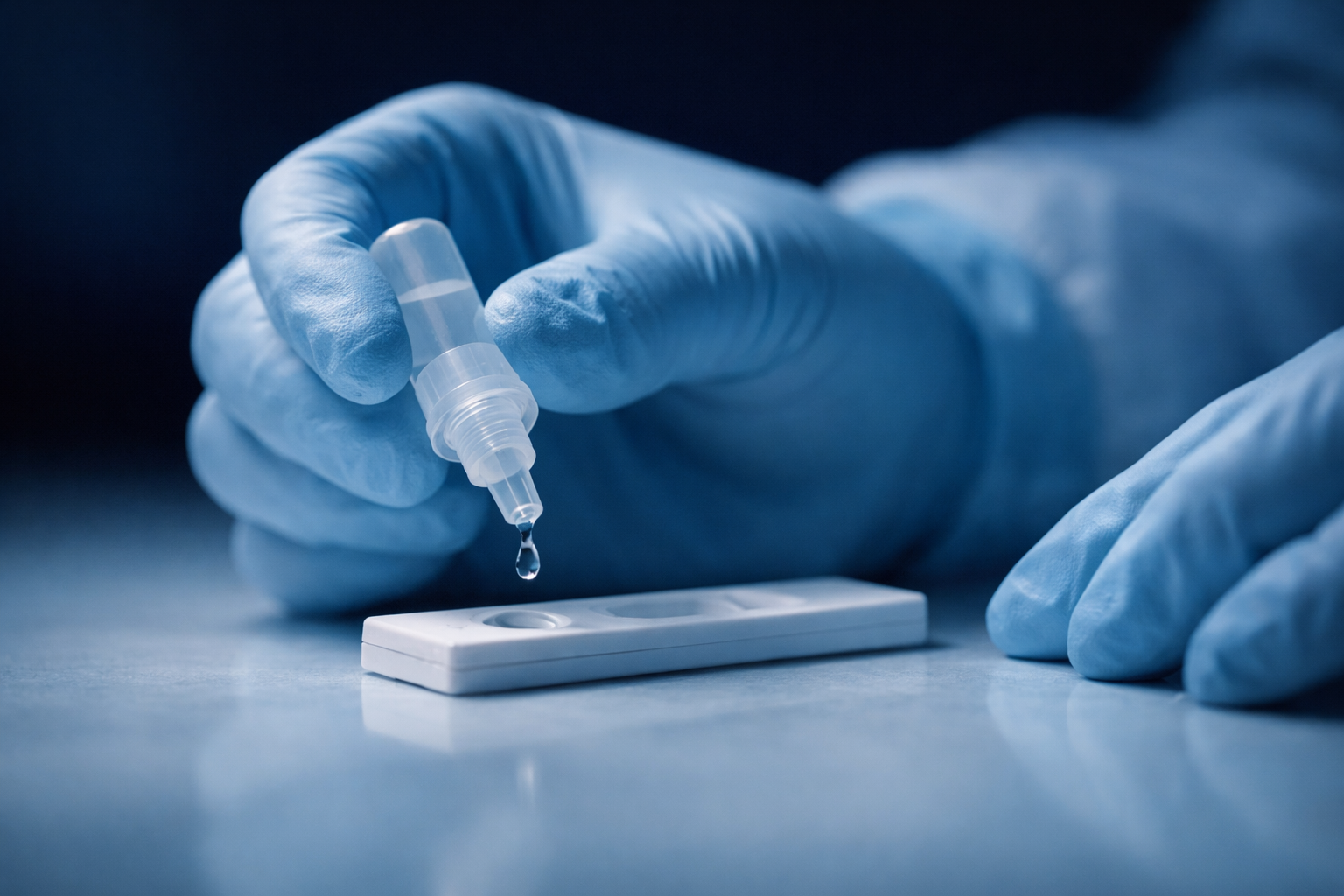
The baseline of drug-checking. Trusted lateral flow detection for the most widespread synthetic opioid in the unregulated supply.
Standard offeringThe only U.S. supplier offering all five emerging-threat drug-checking analytes from a single source. Fentanyl, xylazine, medetomidine, nitazene, benzodiazepines, and any custom multi-panel a program needs.
Diagnostic development partners across 25 years include
The Portfolio
The contemporary drug supply changes faster than most labs can validate a new test. We are the only U.S. supplier offering all five emerging-threat analytes from a single source, configurable into any custom multi-panel a program requires.
The baseline of drug-checking. Trusted lateral flow detection for the most widespread synthetic opioid in the unregulated supply.
Standard offeringThe veterinary tranquilizer now driving the next wave of overdose presentations. Independent benchmarking against competing strips demonstrates substantially improved sensitivity.
High demandAn emerging veterinary tranquilizer increasingly displacing xylazine in regional markets. Detection across racemic mixtures, validated on authentic field samples.
Standard offeringA class of synthetic opioids whose potency can exceed fentanyl by orders of magnitude. U.S.-supplied. Designed to address documented gaps in competing strips, including detection of analogs that incumbent products miss.
Standard offeringDetection across the benzodiazepine class, including the designer analogs increasingly found in counterfeit pressed pills.
Standard offeringWe configure multi-analyte panels for specific use cases: settlement-fund field deployments, syringe service programs, hospital intake, custodial and corrections settings. Tell us what you need to detect.
Custom buildBeyond Drug Checking
Drug-checking is our current focus, not the limit of what we do. The lateral-flow chemistry, supply chain, and regulatory work behind the five-analyte portfolio also sits underneath a broader set of diagnostic capabilities developed across thirty years.

A point-of-care lateral-flow assay for sickle cell screening, designed for non-laboratory deployment in resource-limited settings. Originally developed in response to a request from a national ministry of health for a field-deployable alternative to lab-based electrophoresis.
Lateral-flow immunoassay for the simultaneous detection and differentiation of SARS-CoV-2, Influenza A, and Influenza B antigens from anterior nasal or nasopharyngeal swabs. Visual read in roughly fifteen minutes. CLIA-waived format, FDA EUA where applicable, suited to clinic, urgent care, and turnkey program deployment.
Standard offering
Thirty years of building assays for problems that did not have an existing test. Antibody, molecular, and antigen platforms. Custom multi-panels assembled from any combination of our analytes. Custom packaging, language localization, in-person and online training, regulatory submissions, and temperature-controlled logistics where required.
Project basisWho We Serve
Our supply chain, pricing structure, and technical support are designed for the procurement realities of public-sector and large institutional buyers, not consumer retail.
Procurement against opioid-settlement fund allocations. Bulk pricing, rolling release schedules, training support included.
A U.S.-based supplier with a vetted partner manufacturing network supporting federal contract vehicles including the Federal Supply Schedule (FSS), Distribution and Pricing Agreement (DAPA), and Electronic Catalog (ECAT). DSG holds an active CAGE Code and is registered in the System for Award Management (SAM).
Direct supply to syringe service programs, mobile outreach teams, and state-funded harm reduction networks. Hub-pricing for distribution partnerships.
ED intake, behavioral health, custodial settings. Multi-panel configurations available for specific clinical workflows.
Intake testing and reentry support across state and county jurisdictions. Volume-appropriate pricing and packaging.
Diagnostic deployments at the national level for the governments of seven countries to date. Temperature-controlled logistics available where required.
Why DSG
The only U.S. supplier offering all five emerging-threat drug-checking strips from a single source: fentanyl, xylazine, medetomidine, nitazene, and benzodiazepines. Configurable into any custom multi-panel a program requires.
We work with established diagnostic manufacturers, including U.S. partners such as Princeton BioMeditech (LifeSign) and U.S.-based assay developers, and validate every lot before it ships. Procurement officers get a domestic point of contact, a domestic supply contract, and firm lead times.
The same team that has developed assays for the world's largest pharmaceutical companies, national governments, and the Red Cross. Drug-checking is a continuation of that work, not a pivot away from it.
SAM-registered. CAGE Code assigned by the Defense Logistics Agency. Documentation and contracting infrastructure already in place for federal, state, and county purchasing.
Quote structure built for settlement-fund procurement, GPO pull-through, and multi-jurisdiction roll-outs. Not a retail SKU pretending to be a B2B product.
Drug-checking is a moving target. We follow what is appearing in the supply, advise on panel configurations, and support training for frontline staff. In-person and online programs available.
Our QA program is aligned to the published academic lot-testing framework from the Lieberman group at Notre Dame and to the new AOAC standards (SMPR 2025.002 and SP 2025.001) covering benzodiazepines, fentanyl, medetomidine, nitazenes, and xylazine. Read the methodology →
Our Story
Diagnostic Solutions Group has developed custom diagnostic solutions for thirty years. Our work spans complex mass screening for blood banks, individual point-of-care testing, and large-scale global deployments across antibody, molecular, and antigen platforms.
Our partners have included Johnson & Johnson, Pfizer, Novo Nordisk, Abbott Laboratories, Ortho Clinical Diagnostics, and the Red Cross. We have supported national diagnostic deployments for the governments of Nigeria, Côte d'Ivoire, Bangladesh, Liberia, Jamaica, India, and Mali. Our work has reached numerous Broadway productions, hundreds of hospitals and urgent care centers, and a wide range of long-term care facilities.
There is no one-size-fits-all diagnostic. The right answer depends on the pathogen, the venue, the operator, and the workflow. That has been true for performing-arts COVID screening, for in-office differential testing of COVID against influenza, and for dengue screening in rural settings. It is just as true for the harm-reduction work that defines our current focus.
Today the bulk of our development and supply is concentrated on five drug-checking analytes: fentanyl, xylazine, medetomidine, nitazene, and benzodiazepines. We ship them as single-analyte strips or as custom multi-panels assembled to a buyer's specification. We are the only U.S. supplier offering all five from a single source.
Our QA program is aligned to the published academic lot-testing framework from the Lieberman group at Notre Dame and to the AOAC standards released in late 2025 (SMPR 2025.002 and SP 2025.001), the closest thing the harm-reduction strip market has to a community standard. We document and disclose lot QC data with every institutional order. Read the methodology →
Alongside the harm-reduction portfolio we continue to manufacture and ship a rapid antigen test for COVID-19 and influenza A&B for clinic, urgent-care, and turnkey deployment, and a rapid sickle cell screening assay supported by AI-assisted result interpretation, originally developed at a national ministry of health's request as a field-deployable alternative to lab-based hemoglobin electrophoresis.
Our solutions extend beyond the test itself. We provide test interpretation supported by artificial intelligence, AWS-based data management and insights, custom packaging, language translations, custom inserts, and in-person and online training programs.
We are end-to-end supply chain experts, specializing in the import and export of diagnostic tests and reagents. We obtain regulatory approvals, registrations, clearances, and emergency use authorizations. When the product requires it, we provide temperature-controlled logistics.
We are a registered supplier to the United States Government and active in the System for Award Management (SAM). We hold a Commercial and Government Entity (CAGE) Code from the Defense Logistics Agency.
Latest research
Contact
Procurement inquiries, panel configurations, settlement-fund timelines, and turnkey program design. We respond to every inquiry personally.